chair conformation determination Chair conformation is the term used in organic chemistry that represents the chair-like structure of a carbon ring consisting of six carbon atoms. In a chair there are three carbons that are like mountain peaks red balls and three that are notches b.
Chair Conformation Determination, The lactam ring adopts an envelope conformation whereas the cyclohexane ring has an almost ideal chair conformation. And now the stabilities. Observe if the puckered ring oxygen atom lies above p ve the plane of the ring or below p -ve.
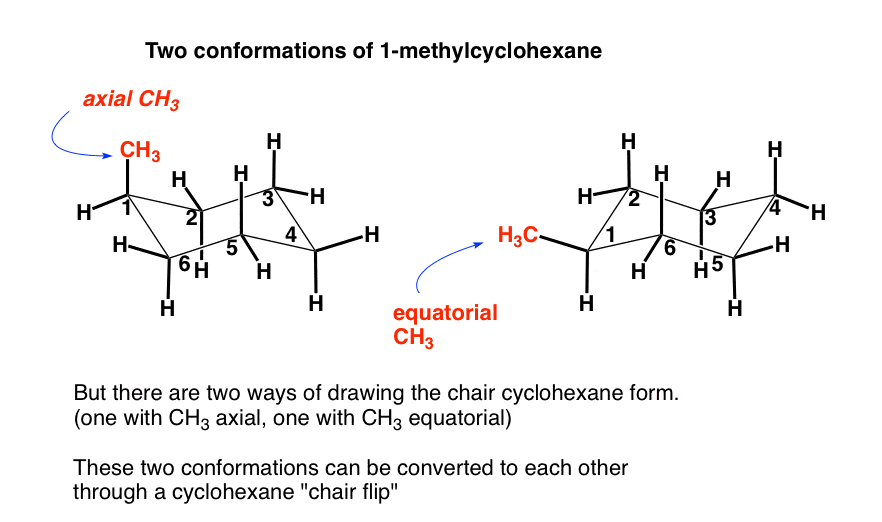 The Cyclohexane Chair Flip Master Organic Chemistry From masterorganicchemistry.com
The Cyclohexane Chair Flip Master Organic Chemistry From masterorganicchemistry.com
An unstable conformation of cyclohexane is the boat conformation which is 71 kcalmol higher in energy than the chair form. H This single bond fixes all of the other bonds in the Newman. The relative stability of substituents is determined by repulsive non- bonded interactions steric compression. Cyclohexane can flip between two chair conformations. As we learned in Chapter 3 configuration relates to the connectivity of atoms.
There are four molecules in the asymmetric unit of γ-spirolactam 1a which show small conformational differences.
In a chair there are three carbons that are like mountain peaks red balls and three that are notches b. Next add a downward-pointing V tip to one end this is the tail of the chair. There are four molecules in the asymmetric unit of γ-spirolactam 1a which show small conformational differences. First thing is to number your substituents or carbon atom on the initial molecule like you did already. Number the ring and draw any chair conformation of the compound.
Another Article :

Cyclohexane is the chair conformation shown below. Active 5 years 6 months ago. Chair conformation chirality. Stretched-Chair Conformation Figures 1 and 2 were the energies determined for the chair conformation. Chair conformation lounge chair - used to kick back and relax. It 4762 kcalmol1 Similarly most mono- and di-substituded is likely that intramolecular hydrogen bonding is not contribut- cyclohexanes exist preferentially in the chair conformation with ing to the preference for chair conformers with axial substituents equatorial substituents12 However for molecules in which large as the crystal. Ring Flipping An Overview Sciencedirect Topics.

A molecule might exist with either the S or R configuration at a chirality center but these two possibilities represent different moleculesthey cannot interconvert without breaking and reforming chemical bonds. Then you should familiarize yourself with the geometry of chair conformation. I cant see how the structure drawn is the enantiomer. The relative stability of substituents is determined by repulsive non- bonded interactions steric compression. Chair conformation is the term used in organic chemistry that represents the chair-like structure of a carbon ring consisting of six carbon atoms. The lactam ring adopts an envelope conformation whereas the cyclohexane ring has an almost ideal chair conformation. Rules Of Thumb Rots For Chair Conformations And Substituent Stability Teach The Mechanism.
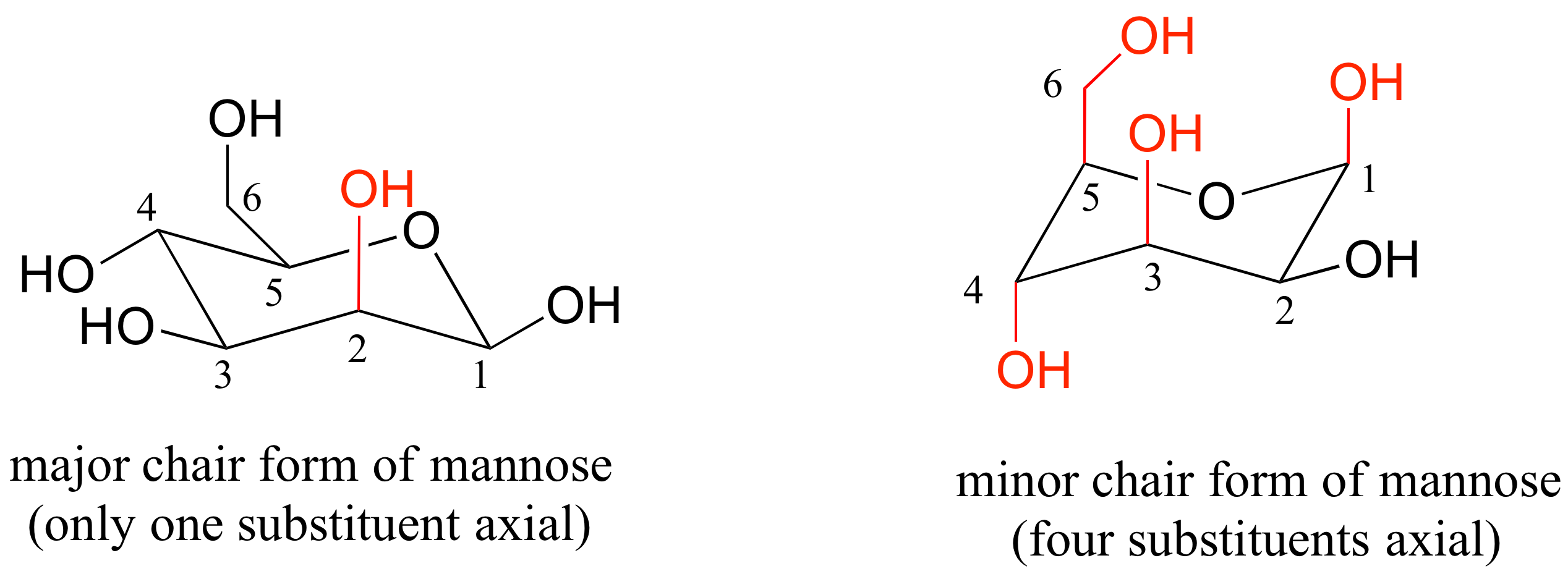
Stretched-Chair Conformation Figures 1 and 2 were the energies determined for the chair conformation. The free energy ΔG of the equilibrium process going from one conformer to the other ie from A to B as shown in step 4 or 5 can be calculated from the equation below where the. The bond lengths within the cyclohexane ring lie in the expected ranges 1514315492 Å whereas one bond to the spiro atom C4 is somewhat longer. From the value of dihedral angle and coupling constant you can identify whether is chair form or boat form. Here we have one chair conformation of methylcyclohexane and this is carbon one you can see we have a methyl group that is axial up at carbon one we also have a hydrogen and Ive made the hydrogen green so we can tell apart from the other hydrogens and this hydrogen is equatorial down at carbon one now this chair conformation is in equilibrium with another chair conformation and we can get to the other chair conformation. If there are no substituents nonhydrogen atoms or groups attached to the ring the two forms will have equal energy. 3 13 Solutions To Chapter 3 Exercises Chemistry Libretexts.

From the value of dihedral angle and coupling constant you can identify whether is chair form or boat form. Ring B has an approximate boat conformation and the bromine atom has the α-configuration. A flip-chair conformation is the conversion of one cyclohexane chair conformation to another by rotation around the carbon-carbon single bonds. Use the steps in Figure 4-28 to practice drawing this chair conformation until you can draw it without having to refer to the figure. The free energy ΔG of the equilibrium process going from one conformer to the other ie from A to B as shown in step 4 or 5 can be calculated from the equation below where the. For each chair conformer add the energy of all the groups on axial position. 0 88 Hz And 0 01 Hz In The 4 Twisted Chair Conformation In Order Download Scientific Diagram.
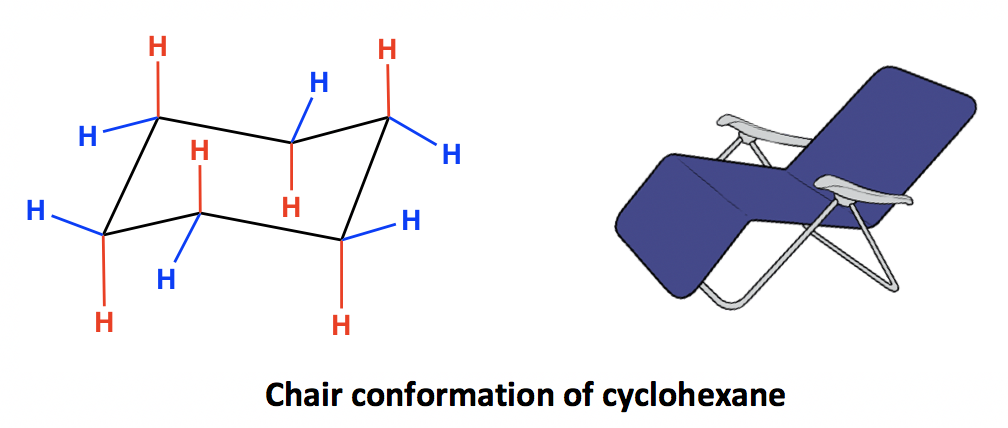
The following guidelines can be used to determine the percent distribution of two chair conformers using the Gibbs free energy equation and some simple algebra. Chem 201Beauchamp Topic 6 Conformations cyclohexanes 2 How to draw a Newman projection of a cyclohexane ring. Chair conformation chirality. Draw the second chair conformation ring-flip-check this post if not sure. Here we have one chair conformation of methylcyclohexane and this is carbon one you can see we have a methyl group that is axial up at carbon one we also have a hydrogen and Ive made the hydrogen green so we can tell apart from the other hydrogens and this hydrogen is equatorial down at carbon one now this chair conformation is in equilibrium with another chair conformation and we can get to the other chair conformation. How do I rotate the ring inversion chair. 4 3 Conformation Analysis Of Cyclohexane Organic Chemistry.

Viewed 2k times 2 begingroup I have two questions regarding this solution. Cyclohexane is the chair conformation shown below. The lactam ring adopts an envelope conformation whereas the cyclohexane ring has an almost ideal chair conformation. To begin start by drawing two lines that are parallel to each other but not perfectly horizontal as shown here. The bond lengths within the cyclohexane ring lie in the expected ranges 1514315492 Å whereas one bond to the spiro atom C4 is somewhat longer. Draw the second chair conformation ring-flip-check this post if not sure. Axial And Equatorial Planes On The Chair Conformation Of Cyclohexane Organic Chemistry I Youtube.

Chair conformation chirality. In II ring A has a chair conformation with a trans AB ring fusion. The following guidelines can be used to determine the percent distribution of two chair conformers using the Gibbs free energy equation and some simple algebra. H This single bond fixes all of the other bonds in the Newman. Boat chair The boat and chair conformations are interconvertable by passing through some very unstable high energy structures called the half-chair and the twist-boat. As we learned in Chapter 3 configuration relates to the connectivity of atoms. Chair Conformations Of Glucose Youtube.
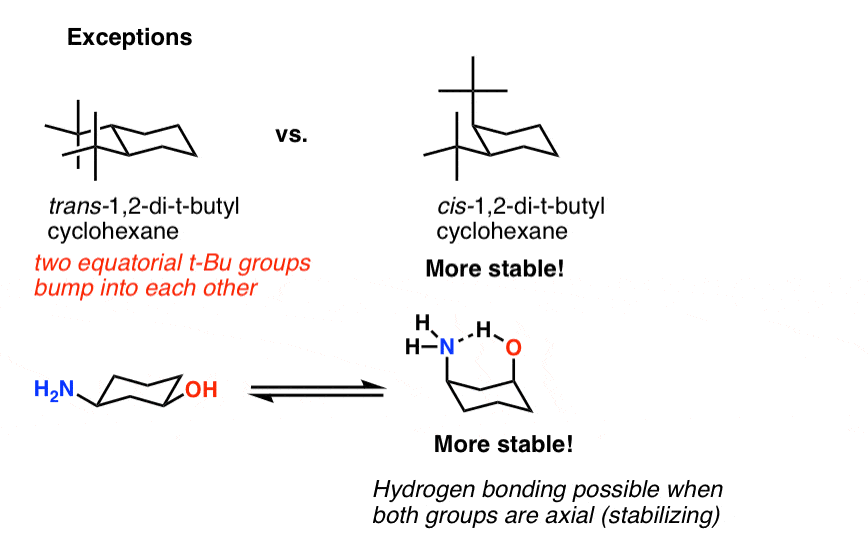
Active 5 years 6 months ago. As we learned in Chapter 3 configuration relates to the connectivity of atoms. Another conformation which is important in any conformational analysis is the transition state or maximum energy conformation on the rotational pathFor cyclohexane this is the so-called half-chair conformation in which now 5 carbons are co-planar and only one is puckered out of the plane. The bromine is in the same configuration in compound III and ring A also adopts the chair conformation. In a chair there are three carbons that are like mountain peaks red balls and three that are notches b. Chair conformation chirality. Cyclohexane Chair Conformation Stability Which One Is Lower Energy.
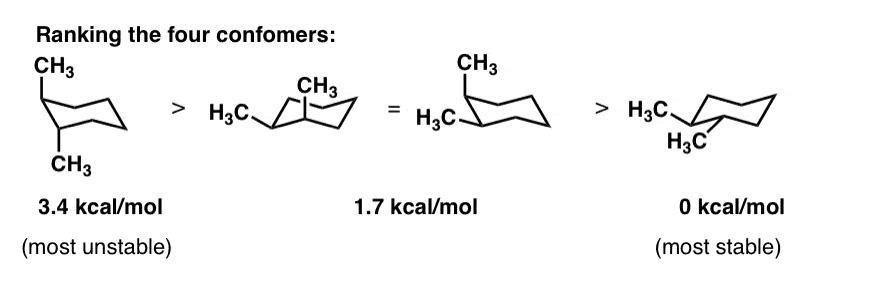
Here we have one chair conformation of methylcyclohexane and this is carbon one you can see we have a methyl group that is axial up at carbon one we also have a hydrogen and Ive made the hydrogen green so we can tell apart from the other hydrogens and this hydrogen is equatorial down at carbon one now this chair conformation is in equilibrium with another chair conformation and we can get to the other chair conformation. From the value of dihedral angle and coupling constant you can identify whether is chair form or boat form. Ring B has an approximate boat conformation and the bromine atom has the α-configuration. And now the stabilities. Specification of Conformation chirality and anomeric form of sugars Determination of chair conformation Locate the anomeric carbon atom and determine if numbering sequence is clockwise n ve or counterclockwise n -ve. A molecule might exist with either the S or R configuration at a chirality center but these two possibilities represent different moleculesthey cannot interconvert without breaking and reforming chemical bonds. Cyclohexane Chair Conformation Stability Which One Is Lower Energy.
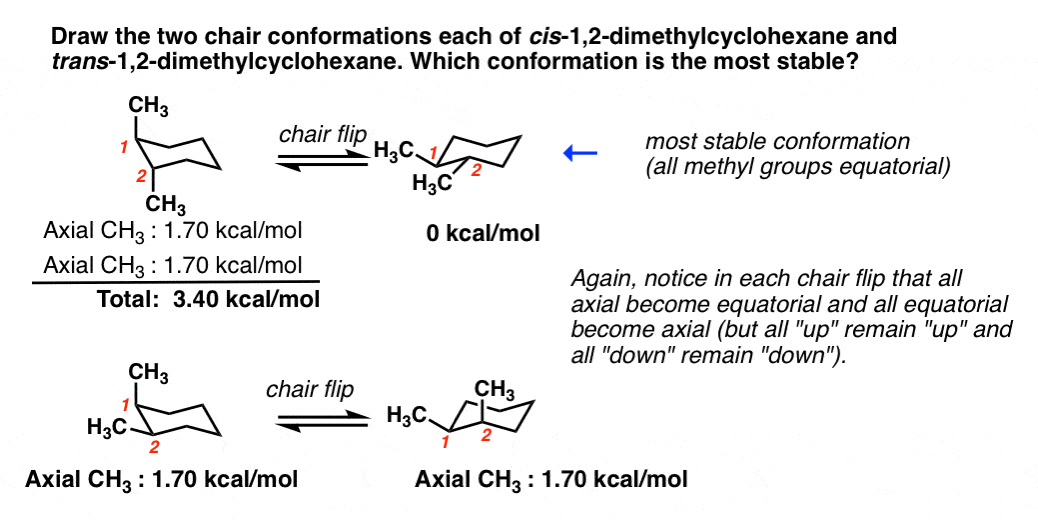
The following chair conformation of cyclohexane is obtained after the one in Figure 4-28 has undergone a chair flip. Now that the molecule is in the boat conformation the energy can be determined for a boat conformation. Boat chair The boat and chair conformations are interconvertable by passing through some very unstable high energy structures called the half-chair and the twist-boat. Active 5 years 6 months ago. There may be the presence of substituents for. Next add a downward-pointing V tip to one end this is the tail of the chair. Cyclohexane Chair Conformation Stability Which One Is Lower Energy.

Active 5 years 6 months ago. Chair conformations are present. The lactam ring adopts an envelope conformation whereas the cyclohexane ring has an almost ideal chair conformation. Bruice Organic Chemistry 6th Edition Chapters 21-215 33-35 51-58 511-513 517 520-5. Boat chair The boat and chair conformations are interconvertable by passing through some very unstable high energy structures called the half-chair and the twist-boat. The free energy ΔG of the equilibrium process going from one conformer to the other ie from A to B as shown in step 4 or 5 can be calculated from the equation below where the. Black Outdoor Balcony Bar Set Zulily Diseno Pequeno Balcon Muebles De Terraza Pequena Diseno De Balcon.
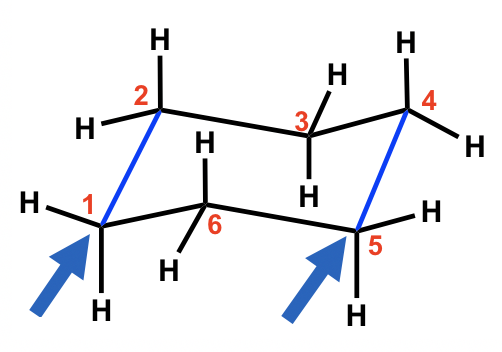
For each chair conformer add the energy of all the groups on axial position. Chair conformations are present. There may be the presence of substituents for. I cant see how the structure drawn is the enantiomer. The free energy ΔG of the equilibrium process going from one conformer to the other ie from A to B as shown in step 4 or 5 can be calculated from the equation below where the. The relative stability of substituents is determined by repulsive non- bonded interactions steric compression. 4 3 Conformation Analysis Of Cyclohexane Organic Chemistry.
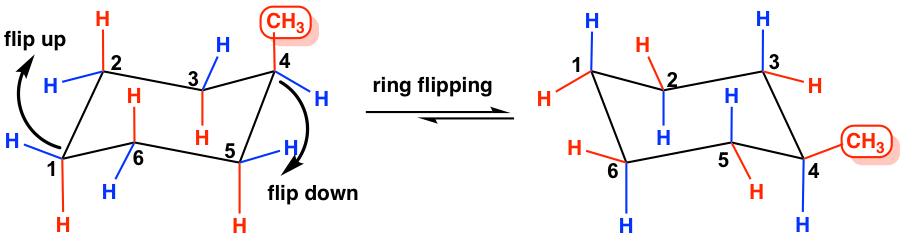
Both compounds adopt chairenvelope conformation. When the chair is flipped the axial positions become equatorial and vice versa. Draw the second chair conformation ring-flip-check this post if not sure. In II ring A has a chair conformation with a trans AB ring fusion. Determined from the name or a 2D drawing. Bruice Organic Chemistry 6th Edition Chapters 21-215 33-35 51-58 511-513 517 520-5. 4 4 Substituted Cyclohexanes Organic Chemistry.

The bond lengths within the cyclohexane ring lie in the expected ranges 1514315492 Å whereas one bond to the spiro atom C4 is somewhat longer. It 4762 kcalmol1 Similarly most mono- and di-substituded is likely that intramolecular hydrogen bonding is not contribut- cyclohexanes exist preferentially in the chair conformation with ing to the preference for chair conformers with axial substituents equatorial substituents12 However for molecules in which large as the crystal. The lactam ring adopts an envelope conformation whereas the cyclohexane ring has an almost ideal chair conformation. An unstable conformation of cyclohexane is the boat conformation which is 71 kcalmol higher in energy than the chair form. How do I rotate the ring inversion chair. Boat chair The boat and chair conformations are interconvertable by passing through some very unstable high energy structures called the half-chair and the twist-boat. Rules Of Thumb Rots For Chair Conformations And Substituent Stability Teach The Mechanism.
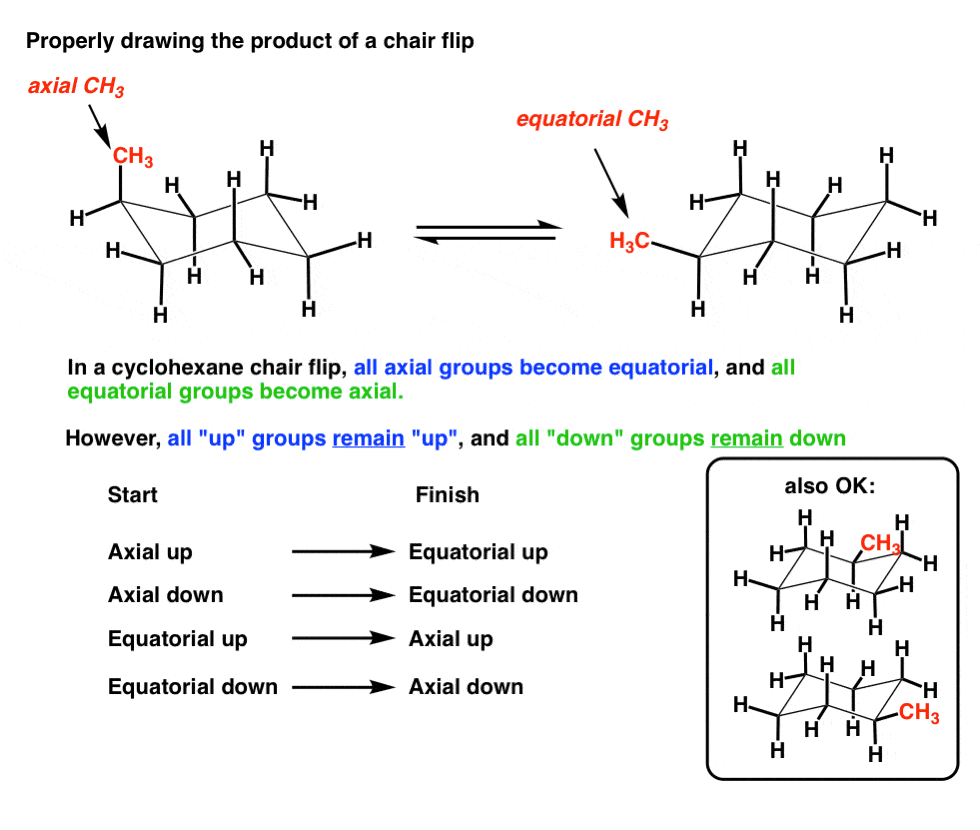
It is important at this stage to clarify the distinction between the terms configuration and conformation. From the value of dihedral angle and coupling constant you can identify whether is chair form or boat form. Then you should familiarize yourself with the geometry of chair conformation. Number the ring and draw any chair conformation of the compound. Finally add an upward-pointing V tip to the other end this is the nose of the chair. If there are no substituents nonhydrogen atoms or groups attached to the ring the two forms will have equal energy. The Cyclohexane Chair Flip Master Organic Chemistry.










